Chemical Ecology · Laboratory Methods
Insect Olfactometry
— A Comprehensive Q&A
Principles, best practices, and scientific context for olfactometer bioassays — with references from the peer-reviewed literature
Contents
Section 1
Fundamentals
What is an insect olfactometer?
An olfactometer is a behavioural assay device used to quantify how insects respond to airborne chemical cues (volatiles or odours). By presenting controlled chemical stimuli and recording an insect's directional movement, entry choices, and dwell time, researchers can determine whether a compound attracts, repels, or otherwise modifies insect behaviour — an essential step in understanding semiochemical communication.
The concept dates at least to 1907, when Barrows documented the first recorded olfactometer experiment using the pomace fly Drosophila ampelophila, and the field has expanded enormously since then, encompassing herbivores, parasitoids, pollinators, and disease vectors.
Why are olfactometers important in chemical ecology?
Olfactometers occupy a foundational position in the pipeline from field observation to applied pest management. They offer a controlled, repeatable laboratory environment where chemical cues can be isolated from visual, tactile, or vibrational stimuli, allowing researchers to attribute insect behavioural responses specifically to volatile compounds. This makes them the first and most critical test before advancing to wind-tunnel studies or field trials.
Practically, olfactometry is indispensable for developing semiochemical-based pest management tools — identifying attractants for traps, repellents for personal protection (e.g. mosquito deterrents), and host volatiles that mediate parasitoid foraging.
What behaviours do olfactometers measure?
Depending on the design, olfactometers can capture several distinct behavioural outputs:
The most commonly recorded metric is binary choice — which arm of a two-arm device the insect enters first — but multi-arm designs allow more nuanced analysis of dwell time and switching frequency. Roberts et al. (2023) note that "four-arm olfactometer bioassay scoring typically involves recording the cumulative amount of time individuals spend in each arm as well as the number of times each arm is entered."
What is the difference between chemotaxis and anemotaxis?
Chemotaxis refers to oriented movement along a chemical concentration gradient — the insect moves toward increasing concentrations of an attractant. Anemotaxis is oriented movement relative to airflow direction — insects fly or walk upwind when they detect an odour plume.
In reality, insects combine both mechanisms in what is called odour-modulated anemotaxis: airflow provides the directional vector and odour detection triggers and sustains the upwind movement. Still-air olfactometers cannot capture anemotaxis, which is a key limitation compared with moving-air designs.
Section 2
Types of Olfactometers
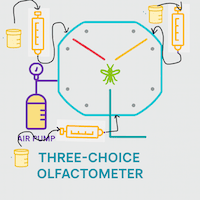
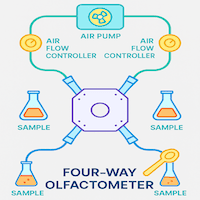
What are the main olfactometer types?
| Type | Airflow | Arms | Best for |
|---|---|---|---|
| Still-air | None | Enclosed arena | Simple proximity assays; low cost |
| Two-arm (Y/T-tube) | Moving | 2 | Binary choice; semiochemical screening |
| Four-arm | Moving (vacuum) | 4 | Small walking insects; dwell-time analysis |
| Six-arm | Moving (vacuum) | 6 | High-throughput screening; multiple odours |
The literature describes three main "moving-air" olfactometer designs in widespread use, first comprehensively categorised by Barrows (1907) and progressively refined through the 20th century (McIndoo 1926; Pettersson 1970; Turlings et al. 2004).
What is a still-air olfactometer, and what are its limitations?
A still-air olfactometer is a simple enclosed arena containing one or more odour sources. The researcher records proximity to the stimulus, time spent near it, or contact events. It was among the earliest olfactometer designs (Barrows, 1907) and remains useful for its low cost and simple setup.
That said, still-air conditions do occur in nature. Lacey & Cardé (2012) showed that some mosquito species locate human-odour sources more effectively in still air than in moving air, so the design is not without ecological merit.
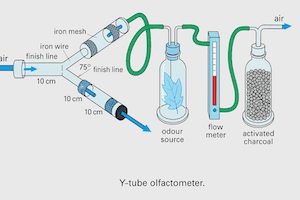
What is a Y-tube olfactometer and how does it work?
A Y-tube olfactometer consists of a main stem that bifurcates into two arms at an angle of 130–150°. Separate airstreams carry the test odour and the control odour through each arm toward the stem. An insect introduced at the base of the stem walks upwind until it reaches the junction and enters one of the two arms — registering a binary choice.
Simultaneous exposure to both airstreams at the junction is the Y-tube's key advantage: the insect experiences both odours at the decision point before committing to a choice, making the comparison ecologically more valid than sequential presentation.
Why prefer a Y-tube over a T-tube?
In a T-tube, the arms are perpendicular (90°) to the stem, meaning the insect must make a sharp turn and may not be simultaneously exposed to both odour streams at the junction. The wider angle of a Y-tube (130–150°) ensures the two airstreams overlap at the decision point, reducing positional and directional bias. The Y-tube design also tends to reduce the influence of the insect's previous direction of travel on its arm choice, improving experimental validity.
What is a four-arm olfactometer?
The four-arm olfactometer was first developed by Hardee et al. (1967) to study the boll weevil, and later refined by Vet et al. (1983) to produce clearly discrete odour fields. A central arena is connected to four arms, each delivering a distinct odour; a vacuum pump draws air inward from each arm tip, maintaining laminar odour fields. The test insect is placed in the central zone where all four odours are detectable before choosing an arm.
A particularly important design consideration: once an insect enters an arm, it can no longer detect the other three odour fields. This means the first entry decision is the most ecologically informative. The standard approach uses one treatment arm and three control arms, setting a 25% probability of choosing the treatment arm by chance alone — a stringent criterion for demonstrating preference.
What is a six-arm olfactometer?
The six-arm olfactometer, first described by Beerwinkle et al. (1996) and refined by Turlings et al. (2004), allows up to six distinct odour sources to be assessed simultaneously. Study organisms released into a central arena can enter any of six horizontal tubes, where they are trapped in a glass bulb for counting. Its principal advantage is throughput — up to six compounds can be screened in a single run, making it ideal for initial compound screening before narrowing down candidates for more detailed two-arm studies.
Section 3
Design & Setup
What materials should olfactometers be made of?
The choice of material is critical because many plastics adsorb volatile compounds, leading to cross-contamination between replicates and potentially altering the odour profile experienced by the insect. The gold standard materials are:
Connecting tubing should likewise be PTFE, and connectors should use brass fittings with PTFE ferrules to avoid reactive metal surfaces (Roberts et al., 2019).
Why is airflow so important, and how should it be prepared?
Airflow serves a dual function: it carries the odour plume to the insect (enabling chemotaxis) and provides the directional vector for anemotaxis. Unequal airflow between arms introduces a systematic directional bias that can produce false positives or mask genuine preferences.
A standard airflow preparation system involves:
- Air pump (or compressed air supply)
- Activated charcoal filter — removes ambient VOCs
- Humidification vessel (deionised water) — prevents desiccation cues
- Flow meters — ensures equal distribution across arms
Airflow rate should be calibrated for the study species: smaller insects require reduced flow to avoid physical disruption of walking behaviour, while higher flow rates may suppress odour plume boundaries (Tichy et al., 2020).
How do you validate airflow and confirm discrete odour fields?
Before any biological trial, airflow patterns should be visualised using a smoke test. One method involves combining the vapours of concentrated hydrochloric acid and ammonia to produce a dense white smoke of ammonium chloride, which can be photographed as it is drawn through the device (Pope, 2004). This confirms that:
Section 4
Experimental Design
How do you test for and eliminate directional bias?
Directional bias occurs when insects preferentially move toward one arm regardless of its odour content — caused by asymmetric light, airflow, or visual cues. It must be tested before any experimental data are collected by running control-versus-control trials: identical stimuli (or clean air) in both arms.
Roberts et al. (2023) recommend: "even where there is no directional bias apparent, to alternate the position of the odour source for each pre-defined replicate."
What environmental conditions must be controlled, and why?
Three environmental parameters consistently affect insect behaviour and must be standardised:
Temperature — Insects are ectotherms: their metabolic rate and locomotory speed are temperature-dependent. Uncontrolled temperature gradients inside an olfactometer can be sensed by thermoreceptors on the antennae, producing responses that mimic (or mask) olfactory preferences (Abram et al., 2017; Budelli et al., 2019).
Humidity — Many insect species detect humidity through hygroreceptors on the distal antennae (Altner & Loftus, 1985). Biological odour sources such as plant leaves release water vapour, meaning insects may respond to the humidity differential rather than the volatiles themselves.
Light — Alternating-current incandescent lamps flicker at 120 cycles per second, well within the 20–300 Hz detectable range of insects (Shields, 1989). This flicker can induce phototaxis that overrides olfactory responses. Fluorescent lamps with electronic ballasts (output ≥40 kHz) or LEDs eliminate this artefact.
Why should visual cues be excluded from olfactometer arenas?
Insects across virtually all major orders use visual cues in addition to olfaction for host location, mate-finding, and predator avoidance. Herbivores respond to plant spectral reflectance (Prokopy & Owens, 1983), parasitoid wasps detect host plant colour (Cochard et al., 2019), and pollinators integrate colour with floral scent (Rachersberger et al., 2019). Any asymmetry in visual stimulation between olfactometer arms constitutes an uncontrolled confound.
The standard mitigation is to wrap the entire olfactometer in opaque material — black or white fabric — and use homogeneous overhead lighting. Where this is not reported in published studies, it becomes impossible to distinguish whether behavioural responses reflect olfactory or visual preferences.
Section 5
Stimulus Preparation
Why is solvent choice important when preparing chemical stimuli?
The solvent determines both the concentration of the test compound and its release rate into the airstream, which change dynamically across the duration of a bioassay. A highly volatile solvent like hexane or diethyl ether produces a burst of high concentration early in the bioassay, which may decline to sub-threshold levels before all replicates are tested. A less volatile carrier like paraffin oil releases the compound more steadily over time, but at lower concentrations that may be insufficient to elicit responses in some species.
What is a common mistake when using biological stimulus material?
Mechanical damage to plant material — snapping stems, crushing leaves, or rough handling — triggers the immediate release of green leaf volatiles (GLVs) such as (Z)-3-hexenol and (Z)-3-hexenyl acetate that are characteristic of wound responses rather than the constitutive or herbivore-induced blend being studied (Dicke et al., 1990). Such inadvertent wounding can completely alter the volatile profile and lead to misleading conclusions about the insect's host-location behaviour.
Biological material should be handled minimally and placed gently into airtight glass chambers with offset air inlets and outlets so that airflow passes uniformly across the entire odour source.
How should controls be chosen for olfactometer experiments?
The choice of control is one of the most consequential — and most frequently flawed — decisions in olfactometer study design. A control should represent the ecological baseline against which the test stimulus is compared.
The appropriate control for an herbivore-attraction study is typically an uninfested plant of the same species, allowing the insect to discriminate between infested and uninfested plant volatiles rather than between volatiles and vacuum. As Kissen et al. (2009) and Roberts et al. (2023) emphasise: "an appropriate control would be an uninfested, non-prey infested, or artificially damaged plant of the same species rather than clean air."
Section 6
Organism Handling
Should insects be tested individually or in groups?
Both approaches are used, but each carries trade-offs. Individual testing avoids intra-group behavioural interactions but is labour-intensive and slow. Group testing increases throughput but introduces the risk of pseudoreplication and social modulation of behaviour.
A notable example of sex-dependent social interference: Turlings et al. (2004) found that female parasitoid wasps did not influence each other's behaviour when released in small groups, but males in mixed-sex groups preferentially oriented toward females rather than the chemical stimulus — a clear social confound that would go undetected without careful preliminary observation.
What is pseudoreplication in olfactometer group testing?
Pseudoreplication occurs when statistically non-independent observations are treated as if they were independent. In an olfactometer releasing groups of ten insects, the movement of insect 3 may be influenced by the presence of insect 7 — they are not independent data points. Treating each of the ten individuals as a separate replicate inflates the apparent sample size and produces falsely narrow confidence intervals.
The correct treatment is to count the entire group release as one replicate, recording the proportion of individuals choosing each arm. This reduces effective sample size but accurately represents data variability.
How does physiological state affect olfactometer results?
Physiological state is one of the most frequently neglected sources of variability in olfactometry. Documented examples include:
For example, Defagó et al. (2016) demonstrated that prior food deprivation significantly increases herbivore responsiveness to host-plant odour cues — a starved insect will appear to "prefer" a host it might ignore when satiated. Saveer et al. (2012) showed that mating switches moth olfactory coding, altering their odour preferences entirely.
Furthermore, behavioural responses vary with the time of day due to circadian rhythms in olfactory sensitivity (Meireles-Filho & Kyriacou, 2013). Bioassays conducted across multiple days should always be performed during the same time window each day.
Section 7
Data Collection
What types of data are collected, and how is behaviour scored?
The type of data collected depends on the olfactometer design:
| Design | Data type | Example metric |
|---|---|---|
| Still air / Y-tube / 6-arm | Binary count | Number choosing each arm |
| Four-arm | Temporal | Seconds spent in each arm; entry count |
Scoring can be done manually with a stopwatch and ethogram, or via dedicated software. Proprietary options include OLFA (Nazzi, 1995) and Noldus Observer (Mizuno et al., 2022); Noldus EthoVision XT enables automated video-tracking (McCormick et al., 2016). Open-source alternatives such as JWatcher are also well-validated. For a comprehensive review of open-source tracking software, Panadeiro et al. (2021) examined 28 platforms and their comparative features.
Section 8
Statistical Analysis
What statistical tests are commonly used for Y-tube data?
Binary count data from two-arm olfactometers are most commonly analysed using a chi-squared goodness-of-fit test or a binomial exact test. Both assess whether the observed distribution of choices deviates from a 50:50 expectation. These tests are valid when the data are truly independent and uncorrelated — conditions that are often not met in practice.
What is the recommended modern statistical approach?
Generalised Linear Mixed Models (GLMMs) are now the recommended framework for olfactometer data. They offer several critical advantages over classical chi-squared or t-tests:
For temporal (dwell-time) data from four-arm olfactometers, the data are compositional — time in one arm directly constrains time in others — requiring a log-ratio transformation before fitting a Gaussian GLMM (Epel, 2013; Aitchison & Egozcue, 2005).
What is statistical power, and why does it matter?
Statistical power is the probability of correctly detecting a real biological effect when one exists. It is determined by sample size, effect size, and the chosen significance threshold (α). A study with insufficient power will fail to detect true preferences (false negative) and waste biological material and researcher effort.
Roberts et al. (2023) argue that "the use of arbitrary sample size in experimental designs is rarely, if ever, appropriate" — yet this is commonly observed in published olfactometer studies where sample sizes appear to have been chosen by convention rather than power analysis. The consequences are not merely statistical: underpowered studies that narrowly miss significance contribute to publication bias when only statistically significant results are published.
A power analysis requires an estimate of the expected effect size, ideally from pilot data or the prior literature, and should be conducted before data collection begins (Cohen, 1992).
Section 9
Cleaning & Maintenance
Why is cleaning so critical, and what protocol should be followed?
Residual volatiles from previous replicates — whether from the odour source itself or from semiochemicals deposited by the study insect (alarm pheromones, contact kairomones, faeces) — are a primary source of cross-contamination that can bias subsequent trials. A rigorous cleaning protocol is not optional; it is integral to scientific validity.
Standard protocol for glass olfactometers (Roberts et al., 2023):
- Soak in dilute fragrance-free detergent (e.g. 5% Decon 75) for 15 min — removes biological residues
- Rinse with warm water
- Rinse with HPLC-grade acetone — dissolves most remaining volatile residues
- Bake in glassware oven at ≥120 °C for ≥15 min — drives off remaining organics
Plastic components cannot withstand acetone or high temperatures; ethanol is preferred, followed by air-drying in a fume hood. PET bags should be treated as single-use and discarded after each trial.
How are activated charcoal filters maintained?
Activated charcoal filters have a finite adsorption capacity. Once saturated, they cease to remove contaminant VOCs from the incoming airstream, potentially exposing study insects to background laboratory odours that confound their behaviour. Filters should be periodically regenerated by heating to 220 °C under a stream of inert nitrogen gas for up to 60 minutes (Dutta et al., 2019; Roberts et al., 2019). Even with regular regeneration, charcoal filters have a finite operational lifespan and should be replaced on a schedule informed by experiment frequency and ambient VOC load.
Section 10
Interpreting Results
What do "attractant" and "repellent" mean, and why can these terms mislead?
The terms "attractant" and "repellent" were formalised by Dethier et al. (1960) to describe chemicals that cause purposeful movement toward or away from a chemical source, respectively. However, Kennedy (1977) cautioned that these are portmanteau concepts — an insect may aggregate near a stimulus not because it is directionally attracted but because an "arrestant" has reduced its movement speed or increased turning rate upon random encounter.
The updated classification framework of Miller et al. (2009) distinguishes between taxis-based responses (kinetic attractant, kinetic repellent, tactic attractant, tactic repellent) and engagement states (engagent vs. disengagent), providing greater precision in describing what is actually observed.
Section 11
Common Pitfalls
What are the most common mistakes in olfactometer studies?
Roberts et al. (2023) synthesise the critical failure points across hundreds of published studies:
| Pitfall | Consequence | Remedy |
|---|---|---|
| Unequal airflow between arms | Systematic directional bias | Flow meters; smoke validation |
| Inappropriate controls | Ecologically meaningless comparisons | Match controls to field conditions |
| Pseudoreplication | Inflated sample size; false significance | GLMMs; treat group as one replicate |
| Uncontrolled environment | Temperature / humidity / light artefacts | Climate-controlled room; LED lighting |
| Inadequate cleaning | Residual odour cross-contamination | Solvent rinse + oven bake protocol |
| Arbitrary sample sizes | Underpowered conclusions | A priori power analysis |
What ensures reproducibility in olfactometer research?
Reproducibility requires that every methodological decision be standardised and reported in sufficient detail for independent replication. Roberts et al. (2023) identify five pillars of reproducibility in olfactometry:
Effect size is particularly important: a statistically significant result with a tiny effect size has limited biological relevance and may be an artefact of large sample sizes. Reporting effect sizes alongside p-values allows meta-analysts to synthesise evidence across studies and guards against publication bias toward marginal significance (Nakagawa & Schielzeth, 2010; Head et al., 2015).
Section 12
Advanced Concepts
What is compositional data, and why does it require special analysis?
Four-arm olfactometer data are inherently compositional: the time an insect spends in arm A necessarily reduces the time available for arms B, C, and D. This creates a mathematical constraint — all proportions sum to 1 — meaning the data points are not independent. Standard statistical tests designed for independent data (t-tests, ANOVA) violate this assumption, typically leading to effect-size underestimation.
What is effect size, and why should it always be reported?
Effect size quantifies the magnitude of a difference — not merely its statistical significance. A large, well-powered study can detect a statistically significant preference where an insect spends 51% of time in the treatment arm vs. 49% in the control arm; this is significant but biologically trivial. Effect sizes such as Cohen's d, odds ratios, or partial η² provide the reader with the information needed to judge biological importance.
Moreover, effect sizes are the currency of meta-analysis: they allow comparison and synthesis of results across studies using different sample sizes and methodologies. Failure to report effect sizes contributes to publication bias, where only large or marginally significant effects are published (Head et al., 2015; Nakagawa & Schielzeth, 2010).
Can olfactometers accurately mimic natural conditions?
Olfactometers offer a controlled simplification of nature, not a faithful reproduction of it. Key differences from the field include: laminar rather than turbulent odour plumes, constrained arenas that prevent long-range orientation, absence of multimodal cues (visual, tactile, magnetic), and insects in potentially non-representative physiological states.
Nevertheless, this simplification is the feature — not the bug. By controlling all variables except the chemical stimulus of interest, olfactometers allow attributing a behavioural response to a specific compound or blend with a confidence impossible to achieve in the field. The appropriate role of olfactometry is hypothesis generation and mechanistic investigation, with findings then tested in ecologically realistic wind-tunnel and field-release experiments.
Scientific References
- Roberts JM, Clunie BJ, Leather SR, Harris WE & Pope TW (2023) Scents and sensibility: Best practice in insect olfactometer bioassays. Entomologia Experimentalis et Applicata 171: 808–820. doi:10.1111/eea.13351
- Ramírez CC, Fuentes-Contreras E, Rodríguez LC & Niemeyer HM (2000) Pseudoreplication and its frequency in olfactometric laboratory studies. Journal of Chemical Ecology 26: 1423–1431.
- Martínez AS & Hardie J (2009) Hygroreception in olfactometer studies. Physiological Entomology 34: 211–216.
- Turlings TCJ, Davison AC & Tamò C (2004) A six-arm olfactometer permitting simultaneous observation of insect attraction and odour trapping. Physiological Entomology 29: 45–55.
- Vet LEM, van Lenteren JC, Heymans M & Meelis E (1983) An airflow olfactometer for measuring olfactory responses of hymenopterous parasitoids and other small insects. Physiological Entomology 8: 97–106.
- Kennedy JS (1977) Behaviorally discriminating assays of attractants and repellents. In: Chemical Control of Behavior: Theory and Application (eds McKelvey JJ & Shorey HH). Wiley Interscience, New York.
- Dethier VG, Browne BL & Smith CN (1960) The designation of chemicals in terms of the responses they elicit from insects. Journal of Economic Entomology 53: 134–136.
- Miller JR, Siegert PY, Amimo FA & Walker ED (2009) Designation of chemicals in terms of the locomotor responses they elicit from insects: an update of Dethier et al. (1960). Journal of Economic Entomology 102: 2056–2060.
- Dicke M, van Beek TA, Posthumus MA et al. (1990) Isolation and identification of volatile kairomone that affects acarine predator-prey interactions: involvement of host plant in its production. Journal of Chemical Ecology 16: 381–396.
- Abram PK, Boivin G, Moiroux J & Brodeur J (2017) Behavioural effects of temperature on ectothermic animals: unifying thermal physiology and behavioural plasticity. Biological Reviews 92: 1859–1876.
- Shields EJ (1989) Artificial light: experimental problems with insects. Bulletin of the Entomological Society of America 35: 40–45.
- Nakagawa S & Schielzeth H (2010) Repeatability for Gaussian and non-Gaussian data: a practical guide for biologists. Biological Reviews 85: 935–956.
- Cohen J (1992) Statistical Power Analysis. Current Directions in Psychological Science 1: 98–101.
- Aitchison J & Egozcue J (2005) Compositional data analysis: where are we and where should we be heading? Mathematical Geology 37: 829–850.
- Head ML, Holman L, Lanfear R, Kahn AT & Jennions MD (2015) The extent and consequences of p-hacking in science. PLoS Biology 13: e1002106.
- Barrows WM (1907) The reactions of the pomace fly, Drosophila ampelophila Loew, to odourous substances. Journal of Experimental Zoology 4: 515–537.
- Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecological Monographs 54: 187–211.
- Saveer AM, Kromann SH, Birgersson G et al. (2012) Floral to green: mating switches moth olfactory coding and preference. Proceedings of the Royal Society B 279: 2314–2322.
- Panadeiro V, Rodriguez A, Henry J, Wlodkowic D & Andersson M (2021) A review of 28 free animal-tracking software applications: current features and limitations. Lab Animal 50: 246–254.
- Brunner M et al. (2025) OlfactionROOM: An optimised, low-cost olfactometer and easy-to-apply setup to mitigate the escape behaviour of insects. Ecological Entomology. doi:10.1111/een.13440