🧪 Standard Operating Procedure (SOP)
Four-Way Olfactometer Assay for Insect Behavior
1. Objective
↑ Back to ContentsTo evaluate insect behavioral responses to multiple odor sources simultaneously using a four-arm olfactometer under controlled airflow and symmetrical conditions.
2. Apparatus & Materials
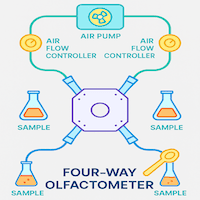
↑ Back to Contents- Four-way olfactometer (cross-shaped arena with central release chamber)
- Air delivery system (pump + 4-channel flow control)
- Activated charcoal filters
- Humidifying chamber (water wash bottles)
- Flow meters (individual for each arm)
- Odor chambers (4 independent sources)
- PTFE/silicone tubing
- Insect collection aspirator
- Stopwatch / video tracking system
- Data recording sheet
3. Experimental Design (VERY IMPORTANT DIFFERENCE vs Y-TUBE)
↑ Back to ContentsUnlike Y-tube:
- Insects do not make a single final choice
- Data is based on:
- Time spent in each arm
- Number of visits
- First entry (optional)
👉 This is a preference distribution assay, not binary choice.
🧪 Conceptual Difference: Y-Tube vs Four-Way Olfactometer
Unlike a Y-tube olfactometer, where insects are forced into a binary decision (choice vs control), a four-way olfactometer allows insects to move freely among multiple odor fields simultaneously. As a result, insects do not make a single irreversible choice, but instead exhibit dynamic, continuous behavioral responses.
What is actually measured?
In four-way olfactometer assays, insect behavior is quantified using:
- Time spent in each arm (residence time)
- Number of visits or entries into each arm
- First arm entered (optional, less robust metric)
These parameters reflect behavioral preference intensity rather than discrete choice.
📊 Why this is NOT a “choice assay”
In a Y-tube:
- The insect commits to one arm → decision is final
- Output = binary data (A vs B)
In a four-arm olfactometer:
- The insect can:
- Enter multiple arms
- Return to the center
- Revisit arms repeatedly
- There is no forced commitment
👉 Therefore, the assay measures:
“Relative preference distribution over time” rather than a single decision event.
🔬 Scientific Basis
This interpretation is well established in chemical ecology:
- Willem Takken & Teun Dekker (1999–2013) — Demonstrated that mosquito responses in multi-port olfactometers are best interpreted using time allocation and movement patterns, not just entry.
- Louise Vet et al. (1983, 1988) — In parasitoid wasp studies, the four-arm olfactometer was specifically designed to measure arrestment and searching behavior, quantified by time spent in odor fields.
- Pettersson (1970s foundational work) — Established that multi-arm olfactometers evaluate orientation and residence behavior, not forced choice.
- Later reviews in chemical ecology confirm that: Residence time in odor zones is a proxy for attraction strength or behavioral arrestment, especially in walking insects.
🧠 Behavioral Interpretation
Each parameter reflects a different biological meaning:
- Time spent in arm → Attraction / arrestment strength
- Number of visits → Exploration vs preference
- Repeated returns → Sustained stimulus engagement
👉 This makes the 4-way olfactometer particularly useful for:
- Subtle odor discrimination
- Dose-response gradients
- Multi-odor comparisons
4. Pre-Experiment Setup
↑ Back to Contents4.1 Cleaning
- Wash with detergent → distilled water → ethanol
- Air dry completely
- Avoid odor carryover
All olfactometer components must be cleaned thoroughly to avoid contamination and odor carryover. For glassware, it is recommended to soak the components overnight in a mild laboratory detergent solution to ensure removal of residual organic compounds. For plasticware, avoid prolonged soaking; instead, immerse in mild soapy water for not more than 15–20 minutes, as extended exposure may lead to surface deposits or adsorption of residues. After soaking, rinse systematically: one rinse with tap water to remove bulk detergent, followed by two rinses with laboratory-grade water, and finally one to two rinses with RO/distilled water to eliminate ionic or particulate contaminants. Where required, perform a final rinse with analytical-grade ethanol to remove volatile residues and accelerate drying. All components should then be air-dried completely in a clean, dust-free environment. Avoid any residual odor contamination, as even trace volatiles can significantly bias insect behavioral responses.
4.2 Airflow Setup
- Equal airflow in ALL 4 arms
- Typical: 200–400 ml/min per arm
- Maintain:
- Laminar flow
- No mixing at center
- Stable pressure
Airflow Setup (Critical and Non-Standard Parameter)
Equal airflow must be maintained in all four arms to ensure symmetry; however, the commonly used range of 200–400 ml/min per arm should not be treated as a fixed standard. The optimal airflow is highly dependent on insect size, behavior, and odor characteristics. For small or weakly mobile insects (e.g., aphids, parasitoids, thrips), even moderate airflow can create mechanical resistance or stress, reducing natural movement and leading to biased results. In contrast, larger or stronger insects (e.g., beetles, moths) may require relatively higher airflow to perceive odor gradients effectively.
From a chemical ecology perspective, airflow directly influences odor plume structure, concentration, and stability. Higher flow rates can dilute semiochemicals and reduce residence time, whereas very low flow may result in odor stagnation or mixing at the केंद्रीय zone. Therefore, airflow must be carefully balanced to achieve laminar flow without turbulence and without cross-arm mixing.
Importantly, several studies (e.g., work by Louise Vet and Teun Dekker) emphasize that behavioral responses are sensitive to both odor concentration and airflow velocity, and that these parameters should be optimized experimentally rather than assumed. Similarly, foundational olfactometer designs by Pettersson highlight that airflow must be adjusted to maintain distinct odor fields while preserving natural insect movement.
👉 Practical recommendation: Airflow should be validated empirically for each experimental system, starting from a moderate baseline and adjusting based on:
- insect mobility and size
- odor volatility and release rate
- absence of turbulence or backflow
- clear behavioral responsiveness
4.3 Odor Placement
- Each arm gets:
- Odor A, B, C, D OR
- 1 treatment + 3 controls
- Rotate odor positions between replicates
👉 Prevents positional bias
In four-way olfactometer assays, each arm can be assigned independent odor sources (e.g., Odor A, B, C, D) or a combination such as one treatment versus multiple controls (e.g., 1 treatment + 3 clean air controls) depending on the experimental objective. Regardless of the design, it is essential to rotate odor positions between replicates to eliminate positional bias arising from subtle asymmetries in airflow, lighting, or apparatus geometry.
This practice is well established in chemical ecology. Studies using four-arm olfactometers (e.g., Vet et al., 1983; Vet et al., 1988) demonstrated that parasitoid responses can be influenced by non-odor cues such as directional airflow or spatial orientation, and therefore recommended systematic rotation of odor sources across arms. Similarly, Pettersson (1970s foundational work) emphasized that even in carefully designed arenas, minor asymmetries can lead to consistent positional preference, necessitating rotation or randomization. More recent mosquito olfactometer studies (e.g., Takken & Knols, 1999; Dekker et al., 2005) also reinforce that randomization and positional switching are critical to avoid bias in multi-port systems.
Common Experimental Designs Used in Literature
Researchers typically adopt one of the following configurations:
1. Full Multi-Odor Comparison (A vs B vs C vs D)
- Used when comparing multiple semiochemicals simultaneously
- Data analyzed as time distribution across arms
- Advantage: high-throughput comparison
- Limitation: interactions between odors possible
2. Single Treatment vs Multiple Controls (1 vs 3)
- One arm contains odor stimulus, remaining arms carry clean air or solvent
- Common in attraction/repellency validation studies
- Provides strong contrast but reduces multi-odor comparison capability
3. Pairwise Testing Within 4-Arm System
- Two arms: treatment vs control
- Remaining arms: blank or duplicates
- Used to improve statistical robustness while maintaining symmetry
4. Dose-Gradient Design
- Same odor at different concentrations in each arm
- Used in dose-response and threshold studies
- Requires careful airflow normalization
5. Replicated Odor Placement (Duplicate Arms)
- Same odor placed in two opposite arms
- Helps test consistency and eliminate directional bias
Why Rotation is Critical
Even in well-built systems, the following can introduce bias:
- Slight differences in tubing length
- Minor airflow variation
- Light gradients
- External environmental cues
👉 Without rotation, insects may show false preference for a position rather than an odor.
Best Practice Recommendation
- Randomize odor positions after every replicate or every few insects
- Ensure each odor appears in all arm positions equally across experiment
- Combine with:
- Control runs (all arms clean air)
- Symmetry checks (equal airflow verification)
- Vet, L.E.M., van Lenteren, J.C., Heymans, M., & Meelis, E. (1983). An airflow olfactometer for measuring olfactory responses of hymenopterous parasitoids and other small insects. Physiological Entomology
- Vet, L.E.M., & Dicke, M. (1992). Ecology of infochemical use by natural enemies in a tritrophic context. Annual Review of Entomology
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of Afrotropical malaria mosquitoes. Annual Review of Entomology
- Dekker, T., Geier, M., & Cardé, R.T. (2005). Carbon dioxide instantly sensitizes female yellow fever mosquitoes to human skin odours. Journal of Experimental Biology
- Pettersson, J. (1970). An aphid olfactometer. Oikos
5. Experimental Conditions
↑ Back to Contents- Temperature: 25 ± 2°C
- RH: 60–80%
- Uniform lighting
- No external odor contamination
Experimental conditions must be carefully controlled to ensure that insect responses are driven primarily by olfactory cues rather than external environmental factors. Typically, assays are conducted at 25 ± 2°C and 60–80% relative humidity, as these ranges support normal insect activity and sensory function. Uniform, diffuse lighting is critical, because many insects exhibit phototaxis (movement toward or away from light), which can strongly influence orientation independent of odor stimuli. Studies have shown that directional or uneven lighting can bias insect movement, leading to false interpretation of olfactory preference (e.g., Kennedy, 1977; Takken & Knols, 1999). Therefore, lighting should be evenly distributed across the arena, avoiding shadows or gradients.
In addition to light, temperature and humidity directly affect insect metabolism, locomotion, and olfactory sensitivity. For instance, olfactory receptor activity and volatile release rates are temperature-dependent, while humidity can influence both insect responsiveness and the dispersion of odor plumes (Dicke & Grostal, 2001; van der Pers & Minks, 1998). Maintaining stable environmental conditions ensures reproducibility and minimizes variability in behavioral responses.
Equally important is the elimination of external odor contamination, as insects are highly sensitive to trace volatiles. Background odors from human presence, chemicals, or laboratory materials can interfere with experimental cues and reduce signal clarity (Vet & Dicke, 1992).
👉 Principle: When evaluating olfactory behavior, all non-olfactory stimuli—such as light gradients, temperature fluctuations, airflow disturbances, and background odors—must be minimized or standardized, so that the observed insect responses accurately reflect true chemical preference rather than environmental bias.
- Kennedy, J.S. (1977). Behavioral mechanisms of orientation to odor sources.
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of Afrotropical malaria mosquitoes. Annual Review of Entomology
- Dicke, M., & Grostal, P. (2001). Chemical detection of natural enemies by arthropods. Annual Review of Entomology
- van der Pers, J.N.C., & Minks, A.K. (1998). Olfactory reception and behavioral responses in insects.
- Vet, L.E.M., & Dicke, M. (1992). Ecology of infochemical use by natural enemies. Annual Review of Entomology
6. Insect Preparation
↑ Back to Contents- Use healthy, active insects
- Standardize:
- Age
- Sex (if needed)
- Feeding status (starved if required)
- Acclimatize before experiment
Insect Preparation (Biological Standardization and Experimental Relevance)
Insect preparation is not merely a handling step but a critical determinant of experimental validity, because insect behavioral responses are tightly regulated by their physiological state, age, circadian rhythm, and ecological context. Therefore, insects used in olfactometer assays must be healthy, active, and biologically aligned with the objective of the experiment, and key parameters such as age, sex, feeding status, and acclimatization must be standardized.
A major factor is age and reproductive status. Insects exhibit age-dependent changes in olfactory sensitivity and behavioral priorities. For example, sex pheromone responsiveness and mate-seeking behavior typically occur only after sexual maturation, and using immature individuals can lead to false negative results. This has been demonstrated in multiple insect systems, where pheromone-mediated responses increase sharply after a species-specific maturation period (Raina et al., 1986; Dickens, 1989; Wyatt, 2014). Thus, if the experimental objective is to study mating preference or pheromone attraction, the test insects must be at the appropriate reproductive stage.
Similarly, sex of the insect must be considered, as males and females often respond differently to the same odor cues. For instance, in many species, males respond to sex pheromones, while females respond more strongly to host or oviposition cues (Takken & Knols, 1999; Bruce et al., 2005). Mixing sexes without control can obscure meaningful behavioral patterns.
Another critical parameter is feeding status. Hunger significantly modulates olfactory-driven behavior; starved insects typically show increased attraction to host or food-related odors, while recently fed individuals may exhibit reduced responsiveness (Dethier, 1982; Simpson & Raubenheimer, 2012). Therefore, standardizing feeding conditions (e.g., starvation for a defined period) ensures consistent motivation across individuals.
Circadian rhythm and activity period are equally important. Many insects exhibit strong diurnal or nocturnal activity patterns, and their olfactory sensitivity is synchronized with these rhythms. Conducting experiments outside the insect's peak activity window can result in reduced movement, delayed responses, or complete inactivity (Saunders, 2002; Bloch et al., 2013). For example, nocturnal moths may show minimal response during daytime assays, even when odor cues are present. Thus, experiments must be aligned with the natural behavioral timing of the species.
The nature of the odor source itself must also match the ecological context. When testing plant-insect interactions, it is important to recognize that different plant parts (leaves, flowers, roots) emit distinct volatile profiles, and these profiles can change with plant age, damage status, or developmental stage (Dicke & Baldwin, 2010; Bruce & Pickett, 2011). Using a whole plant without considering these variations may mask specific behavioral responses. Therefore, researchers often compare whole plant vs individual plant parts vs synthetic blends to accurately interpret insect preference.
In addition, sample size and replication must be sufficient to account for natural behavioral variability. Behavioral assays inherently show high inter-individual variation, and reliable conclusions typically require 20–50 insects per treatment with multiple replicates, as recommended in entomological bioassay standards (Vet et al., 1983; van Lenteren et al., 2003).
Finally, insects should be acclimatized to laboratory conditions prior to testing, allowing them to recover from handling stress and adjust to experimental temperature, humidity, and lighting. Stress or sudden environmental shifts can suppress normal behavior and introduce variability.
Key Principle
In olfactometer experiments, insect response is not only a function of the odor stimulus but also of the biological state of the insect. Proper alignment of experimental design with insect bionomics and ecology is essential to obtain meaningful and reproducible results.
- Wyatt, T.D. (2014). Pheromones and Animal Behavior. Cambridge University Press
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of mosquitoes. Annual Review of Entomology
- Bruce, T.J.A., Wadhams, L.J., & Woodcock, C.M. (2005). Insect host location: a volatile situation. Trends in Plant Science
- Dicke, M., & Baldwin, I.T. (2010). The evolutionary context for herbivore-induced plant volatiles. Trends in Plant Science
- Bruce, T.J.A., & Pickett, J.A. (2011). Perception of plant volatile blends by herbivorous insects. Annual Review of Entomology
- Dethier, V.G. (1982). Mechanisms of host-plant recognition. Entomologia Experimentalis et Applicata
- Simpson, S.J., & Raubenheimer, D. (2012). The Nature of Nutrition.
- Saunders, D.S. (2002). Insect Clocks. Elsevier
- Bloch, G. et al. (2013). Social insect circadian rhythms. Annual Review of Entomology
- Vet, L.E.M. et al. (1983). An olfactometer for behavioral studies. Physiological Entomology
- van Lenteren, J.C. et al. (2003). Quality control in biological control agents.
7. Procedure
↑ Back to ContentsStep 1: Start airflow
- Run system for 2–5 minutes
Step 2: Release insect
- Place insect in central chamber
Step 3: Observation
- Allow free movement in arena
- Record movement continuously
The experimental procedure in a four-way olfactometer is designed to ensure that insects respond to stable, well-defined odor fields under controlled airflow conditions, and that their behavior is recorded without external disturbance.
Step 1: Start Airflow (Pre-conditioning Phase)
Before introducing the insect, the airflow system should be run for 2–5 minutes to allow the formation of stable and discrete odor plumes within each arm. This step is critical because odor transport in olfactometers depends on laminar airflow and steady-state diffusion, and immediately after switching on the system, transient turbulence and uneven odor distribution may occur.
Studies on olfactometer design (e.g., Vet et al., 1983; Pettersson, 1970) emphasize that insects respond to consistent odor gradients, and unstable airflow can lead to ambiguous or non-reproducible behavior. Similarly, work on mosquito and parasitoid olfaction (Takken & Knols, 1999; Dekker et al., 2005) shows that odor plume structure must be stabilized before testing, as insects rely on continuous chemical gradients for orientation.
👉 Therefore, this pre-run period ensures:
- Uniform odor delivery in all arms
- Elimination of turbulence and pressure fluctuations
- Establishment of reproducible experimental conditions
Step 2: Release Insect (Neutral Introduction Zone)
The insect is introduced into the central chamber, which functions as a neutral zone free from directional bias. This ensures that the insect begins the assay without prior exposure to a dominant odor gradient and can sample all available odor fields equally.
Behavioral studies have shown that the initial position of the insect can strongly influence its response; hence, a central release point is essential for unbiased orientation (Kennedy, 1977; Vet & Dicke, 1992). Insects naturally perform klinotaxis and tropotaxis (gradient sampling behaviors), and starting from the center allows them to detect and compare odor cues from multiple दिशाओं.
👉 This step ensures:
- Equal access to all odor sources
- Elimination of positional advantage
- Natural orientation behavior
Step 3: Observation (Continuous Behavioral Recording)
After release, the insect is allowed to move freely within the arena, and its behavior is recorded continuously over the defined observation period. Unlike binary-choice systems, four-way olfactometers capture dynamic behavioral patterns, including movement, residence time, and repeated entries into odor zones.
Continuous observation is essential because insect responses are not instantaneous decisions but evolving behavioral processes, influenced by stimulus strength, internal state, and sensory feedback. Research in chemical ecology demonstrates that time spent in odor zones is a robust indicator of attraction or arrestment (Vet et al., 1983; Bruce et al., 2005).
Advanced studies often use video tracking systems to quantify:
- Time spent in each arm
- Number of visits
- Movement trajectories
👉 Continuous recording ensures:
- Capture of full behavioral response (not just first contact)
- Identification of subtle preferences
- Accurate quantitative analysis
Key Principle
The procedure is structured to ensure that insect behavior reflects true olfactory perception under stable and unbiased conditions, rather than transient airflow effects or positional artifacts.
- Pettersson, J. (1970). An aphid olfactometer. Oikos
- Vet, L.E.M., van Lenteren, J.C., Heymans, M., & Meelis, E. (1983). An airflow olfactometer for measuring olfactory responses. Physiological Entomology
- Vet, L.E.M., & Dicke, M. (1992). Ecology of infochemical use. Annual Review of Entomology
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of mosquitoes. Annual Review of Entomology
- Dekker, T., Geier, M., & Cardé, R.T. (2005). CO₂ sensitization in mosquitoes. Journal of Experimental Biology
- Kennedy, J.S. (1977). Behavioral mechanisms of orientation to odor.
- Bruce, T.J.A., Wadhams, L.J., & Woodcock, C.M. (2005). Insect host location: a volatile situation. Trends in Plant Science
8. Data Recording (CORE SECTION)
↑ Back to ContentsAccurate data recording in a four-way olfactometer is essential because the system measures continuous behavioral preference rather than a single decision event. Therefore, observations must capture spatial distribution and temporal dynamics of insect movement under controlled odor fields.
8.1 Zones Defined
- Central neutral zone
- 4 arm zones (equal size)
The olfactometer arena is divided into a central neutral zone and four symmetrically arranged arm zones, each representing a distinct odor field. The central zone is designed to be odor-balanced, allowing insects to sample odor gradients before committing to any दिशा. Equal sizing of arm zones is critical to ensure comparability of time-based measurements across treatments.
Studies using multi-arm olfactometers (e.g., Vet et al., 1983) emphasize that spatial symmetry is essential to avoid geometric bias, while Pettersson (1970) highlighted the importance of clearly defined zones for interpreting insect orientation behavior.
8.2 What is recorded?
✔️ Primary parameters:
- Time spent in each arm (seconds)
- Number of entries into each arm
✔️ Secondary:
- First arm entered
- Latency to first movement
The primary metrics in four-way olfactometer assays are residence time and visit frequency, as these directly reflect the insect's behavioral preference and arrestment response. Time spent in an odor field is widely accepted as a quantitative proxy for attraction strength, particularly in walking insects and parasitoids (Vet et al., 1983; Vet & Dicke, 1992).
The number of entries provides additional insight into exploratory behavior versus sustained preference, helping distinguish between random movement and true attraction.
Secondary parameters such as first arm entered and latency to movement can be informative but are considered less robust, as they may be influenced by initial orientation bias or stochastic movement (Kennedy, 1977).
More advanced studies, particularly in mosquito and host-seeking research (Takken & Knols, 1999; Dekker et al., 2005), often combine these parameters with trajectory tracking to obtain a complete behavioral profile.
8.3 What is NOT considered?
- Crossing central junction alone = ❌ NOT meaningful
- Brief entry (<2–3 sec) = ❌ often ignored
In four-arm systems, simply crossing the central zone or briefly entering an arm is not considered a valid behavioral response, as insects often perform sampling or probing movements before making a meaningful interaction with an odor field.
Short-duration entries (typically <2–3 seconds) are frequently excluded because they may represent random movement or exploratory scanning rather than true attraction. This approach is supported by behavioral studies showing that insects use sequential sampling strategies (klinotaxis/tropotaxis) before committing to a stimulus (Kennedy, 1977; Vet & Dicke, 1992).
Excluding such transient movements improves signal-to-noise ratio in the data and ensures that recorded responses reflect intentional behavioral engagement.
8.4 Valid behavioral signal
✔️ Insect:
- Enters arm
- Stays for measurable duration
👉 Indicates attraction or arrestment
A valid behavioral response in a four-way olfactometer is defined by entry into an arm followed by sustained residence, indicating that the insect is responding to the odor stimulus. This sustained presence reflects either:
- Attraction (directed movement toward odor source)
- Arrestment (reduced movement due to stimulus retention)
The concept of arrestment behavior is particularly important in multi-arm olfactometers and has been extensively described in parasitoid and herbivore studies (Vet et al., 1983; Bruce et al., 2005). In such cases, insects may not simply move toward an odor but may remain within an odor field, increasing residence time as a behavioral response.
Thus, time spent in an arm is considered one of the most reliable indicators of olfactory preference, especially when compared across multiple odor sources under symmetrical conditions.
- Pettersson, J. (1970). An aphid olfactometer. Oikos
- Vet, L.E.M., van Lenteren, J.C., Heymans, M., & Meelis, E. (1983). An airflow olfactometer for measuring olfactory responses. Physiological Entomology
- Vet, L.E.M., & Dicke, M. (1992). Ecology of infochemical use. Annual Review of Entomology
- Kennedy, J.S. (1977). Behavioral mechanisms of orientation to odor sources
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of mosquitoes. Annual Review of Entomology
- Dekker, T., Geier, M., & Cardé, R.T. (2005). CO₂ sensitization in mosquitoes. Journal of Experimental Biology
- Bruce, T.J.A., Wadhams, L.J., & Woodcock, C.M. (2005). Insect host location: a volatile situation. Trends in Plant Science
9. Observation Time
↑ Back to Contents- Standard: 5–10 minutes per insect
Rules:
- Record entire duration
- Do NOT stop at first entry
The observation period in four-way olfactometer assays is typically set between 5–10 minutes per insect, which allows sufficient time for insects to explore multiple odor fields, perform orientation behavior, and exhibit stable preference patterns. Unlike binary-choice systems, responses in multi-arm olfactometers are dynamic and time-dependent, requiring continuous observation to capture the full behavioral profile.
Recent studies in insect olfaction and host-seeking behavior emphasize that insect responses are not instantaneous decisions but iterative processes involving exploration, sampling, and re-evaluation of odor cues. For example, work by Teun Dekker and colleagues demonstrates that insects such as mosquitoes continuously integrate sensory input over time, with repeated entries and variable residence durations contributing to final behavioral outcomes. Similarly, research on parasitoids and herbivorous insects shows that residence time increases as insects confirm the suitability of an odor source, reflecting a process of behavioral arrestment rather than a one-time choice.
Stopping the experiment at the first arm entry is therefore inappropriate, as this may only represent initial exploration or random movement rather than true preference. Studies in chemical ecology (e.g., Bruce et al., 2015; van Breugel et al., 2015; Cardé & Willis, 2008) highlight that insects often exhibit multi-step orientation behavior, including upwind movement, crosswind casting, and repeated sampling before stabilizing in a preferred odor zone. Continuous recording over the full observation period ensures that these behaviors are captured and quantified accurately.
Moreover, recent advances using video tracking and automated behavioral analysis have reinforced that time allocation across odor fields is a more robust metric than first-choice responses, particularly in complex or multi-odor environments (Gomez-Diaz et al., 2018; van Breugel & Dickinson, 2014). These approaches confirm that meaningful behavioral patterns emerge over time, not at a single decision point.
👉 Practical implication: A 5–10 minute observation window balances:
- Sufficient exploration time
- Stable behavioral response development
- Practical throughput in experimental design
- van Breugel, F., Riffell, J., Fairhall, A., & Dickinson, M.H. (2015). Mosquitoes use vision to associate odor plumes with thermal targets. Current Biology
- Cardé, R.T., & Willis, M.A. (2008). Navigational strategies used by insects to find distant wind-borne sources of odor. Journal of Chemical Ecology
- Bruce, T.J.A., et al. (2015). Odor perception and integration in insect host location. Current Opinion in Insect Science
- Gomez-Diaz, C., et al. (2018). Neural circuits underlying olfactory-driven behavior. Current Opinion in Neurobiology
- van Breugel, F., & Dickinson, M.H. (2014). Plume-tracking behavior of flying insects. Journal of Experimental Biology
- Dekker, T., et al. (multiple studies, 2000–2015) Dynamic odor-guided behavior in mosquitoes
10. Replication
↑ Back to Contents- Minimum: 20–40 insects per treatment
- Multiple experimental runs
Replication (Statistical Reliability and Biological Variability)
Minimum: 20–40 insects per treatment. Multiple experimental runs.
Replication is essential in olfactometer experiments because insect behavior is inherently variable and influenced by both internal (physiological state) and external (micro-environmental) factors. Even under controlled conditions, individual insects may show different levels of activity, responsiveness, or random movement, making single observations unreliable. Therefore, using an adequate number of insects ensures that the observed response reflects a true behavioral trend rather than individual variability.
Behavioral ecology studies consistently emphasize that sample size directly affects statistical power and confidence. With small sample sizes, results may appear biased or inconsistent, whereas increasing the number of insects reduces random error and allows detection of significant differences between treatments. Foundational work in olfactometer bioassays (Vet et al., 1983; Vet & Dicke, 1992) and later methodological reviews highlight that replication across individuals is required to obtain reproducible behavioral patterns.
In addition to biological replication (number of insects), temporal replication (multiple experimental runs) is equally important. Running experiments across different batches or days helps account for day-to-day variation in environmental conditions, insect vigor, or odor release rates. This ensures that results are robust and not dependent on a single experimental condition.
How Confidence is Added to Data
Confidence in olfactometer data is built through a combination of replication, statistical analysis, and validation controls:
- Sufficient sample size (n = 20–40 insects or more): Reduces individual variation and improves reliability
- Consistent trends across replicates: Similar results observed in repeated runs indicate robustness
- Statistical testing: Methods such as Chi-square (for choice data) or ANOVA/Kruskal–Wallis (for time-based data) determine whether observed differences are statistically significant (p < 0.05)
- Control experiments: Running blank tests (e.g., clean air vs clean air) should produce equal distribution, confirming absence of bias
- Reporting variability: Presenting results as mean ± standard deviation (or standard error) reflects the spread of data and increases transparency
- Exclusion or reporting of non-responders: Ensures that inactive insects do not distort results
Scientific Basis
- Behavioral responses in insects show high inter-individual variability, requiring adequate replication for reliable inference (Bell, 1991; Sokal & Rohlf, 1995)
- Chemical ecology studies emphasize the need for replicated assays to distinguish signal from noise (Vet & Dicke, 1992)
- Modern behavioral analysis frameworks highlight that statistical confidence emerges from both sample size and repeatability (Quinn & Keough, 2002)
11. Bias Control
↑ Back to Contents- Rotate odor arms after each replicate
- Clean chamber regularly
- Run control (all arms clean air)
Expected:
- Equal distribution (~25% per arm)
Controlling bias in olfactometer experiments is essential because insect behavior can be influenced not only by odor stimuli but also by subtle asymmetries in airflow, الضوء (light), chamber geometry, or residual chemical cues. Without proper controls, insects may show apparent preference for a location rather than the odor itself, leading to incorrect conclusions.
Rotation of Odor Arms
Rotating odor positions after each replicate is a widely accepted method to eliminate positional bias. Even in well-designed systems, slight differences in airflow resistance, tubing length, or illumination can create consistent directional preference.
In agricultural entomology, studies on parasitoid wasps using four-arm olfactometers (Vet et al., 1983; Vet & Dicke, 1992) routinely rotated odor sources to ensure that host plant volatiles were not confounded with spatial cues. Similarly, research on herbivore responses to plant odors (Bruce et al., 2005; Dicke & Baldwin, 2010) emphasizes randomization of odor placement to avoid systematic bias.
In medical entomology, mosquito studies (e.g., Takken & Knols, 1999; Dekker et al., 2005) also incorporate randomization or rotation of odor ports, as mosquitoes are highly sensitive to environmental gradients and may respond to airflow direction or CO₂ distribution rather than the intended odor stimulus.
Regular Cleaning of Chamber
Regular cleaning of the olfactometer chamber is essential to prevent carryover of semiochemicals, which can persist on surfaces and influence subsequent trials. Many volatile compounds used in insect studies, including plant volatiles and pheromones, can adsorb onto glass or plastic surfaces and be slowly released, creating unintended background signals.
In agricultural systems, experiments with plant volatiles (e.g., Dicke & Grostal, 2001) highlight that residual odors can alter parasitoid behavior, while in mosquito research, even trace contamination can affect host-seeking responses (Cardé & Willis, 2008). Therefore, cleaning between treatments ensures that each assay begins under neutral baseline conditions.
Control Experiments (All Arms Clean Air)
Running control experiments with all arms containing clean air is a fundamental step to verify that the system is free from inherent bias. In an ideal setup, insects should distribute randomly across all four arms (~25% each) in the absence of odor cues.
This approach has been consistently used across disciplines:
- In parasitoid and plant-insect interaction studies (Vet et al., 1983), equal distribution in control runs confirms symmetry of airflow and arena design.
- In mosquito olfaction studies (Takken & Knols, 1999), clean-air controls are used to validate that no directional bias exists before introducing host odors.
If the distribution deviates significantly from the expected 25% per arm, it indicates system bias, which may arise from:
- Unequal airflow
- Light gradients
- Residual odor contamination
- Structural asymmetry
Such issues must be corrected before proceeding with experimental treatments.
Scientific Basis for Bias Control
- Insects integrate multiple sensory cues (odor, airflow, light), and non-olfactory cues can override chemical signals (Kennedy, 1977)
- Randomization and replication are essential to separate true stimulus effects from environmental artifacts (Sokal & Rohlf, 1995)
- Behavioral assays require symmetry and neutrality of the test arena to ensure valid interpretation (Vet & Dicke, 1992)
- Vet, L.E.M. et al. (1983). An airflow olfactometer for behavioral studies. Physiological Entomology
- Vet, L.E.M., & Dicke, M. (1992). Ecology of infochemical use. Annual Review of Entomology
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of mosquitoes. Annual Review of Entomology
- Dekker, T., Geier, M., & Cardé, R.T. (2005). CO₂ sensitization in mosquitoes. Journal of Experimental Biology
- Bruce, T.J.A., Wadhams, L.J., & Woodcock, C.M. (2005). Insect host location. Trends in Plant Science
- Dicke, M., & Baldwin, I.T. (2010). Plant volatile ecology. Trends in Plant Science
- Cardé, R.T., & Willis, M.A. (2008). Odor plume navigation. Journal of Chemical Ecology
- Kennedy, J.S. (1977). Behavioral mechanisms of orientation to odor
- Sokal, R.R., & Rohlf, F.J. (1995). Biometry
12. Data Analysis
↑ Back to Contents12.1 Basic Output
- Mean time per arm
- % time distribution
- Visit frequency
12.2 Statistical Tests
- ANOVA (preferred for 4-arm data)
- Kruskal–Wallis (non-parametric)
- Post-hoc comparisons
12.3 Optional
- Chi-square (for first choice only)
13. Acceptance Criteria
↑ Back to Contents- ✔️ Control = equal distribution
- ✔️ Low inactivity (<30%)
- ✔️ Consistent trends across replicates
14. Cleaning Between Runs
↑ Back to Contents- After 3–5 insects → flush air
- After each treatment → full cleaning
Cleaning Between Runs (Preventing Cross-Contamination and Ensuring Throughput)
After 3–5 insects → flush air. After each treatment → full cleaning.
Cleaning between runs is a critical requirement in olfactometer experiments, as insects are highly sensitive to trace levels of volatile compounds, and even minimal residue from previous assays can significantly bias behavioral responses. Residual semiochemicals can adsorb onto glass or tubing surfaces and be released gradually over time, leading to cross-contamination between treatments. This phenomenon has been well documented in chemical ecology, where carryover effects can alter insect orientation and reduce the reliability of results (Vet et al., 1983; Vet & Dicke, 1992; Bruce et al., 2005).
Flushing the system with clean air after every few insects helps remove transient odor buildup, but it is not sufficient to eliminate adsorbed compounds, especially when working with plant volatiles, pheromones, or high-affinity semiochemicals. Therefore, a complete cleaning of glassware between treatments is essential to restore baseline conditions. Studies in olfactory bioassays emphasize that failure to adequately clean olfactometer components can result in false attraction or repellency responses due to residual odor cues (Dicke & Grostal, 2001; Cardé & Willis, 2008).
From a practical standpoint, this requirement has a direct impact on experimental efficiency. Since proper cleaning involves washing, rinsing, and complete drying, it can introduce significant downtime between runs. To maintain throughput and avoid delays, it is strongly recommended to use multiple sets of glassware, allowing one set to be cleaned and dried while another is in use. This approach is commonly adopted in high-throughput behavioral laboratories to ensure continuous experimentation without compromising data quality.
Scientific Basis
- Volatile compounds can adsorb and desorb from surfaces, affecting subsequent assays (Bruce et al., 2005)
- Insects respond to extremely low concentrations of odors, making contamination a major concern (Takken & Knols, 1999)
- Proper cleaning is essential to maintain experimental reproducibility and signal clarity (Vet & Dicke, 1992)
Practical Recommendation
👉 To ensure both data integrity and experimental efficiency:
- Always perform full cleaning between treatments
- Use air flushing only as an interim step
- Maintain multiple sets of olfactometer glassware to avoid downtime
- Vet, L.E.M. et al. (1983). An airflow olfactometer for behavioral studies. Physiological Entomology
- Vet, L.E.M., & Dicke, M. (1992). Ecology of infochemical use. Annual Review of Entomology
- Bruce, T.J.A., Wadhams, L.J., & Woodcock, C.M. (2005). Insect host location: a volatile situation. Trends in Plant Science
- Dicke, M., & Grostal, P. (2001). Chemical detection of natural enemies. Annual Review of Entomology
- Cardé, R.T., & Willis, M.A. (2008). Navigational strategies of insects. Journal of Chemical Ecology
- Takken, W., & Knols, B.G.J. (1999). Odor-mediated behavior of mosquitoes. Annual Review of Entomology
15. Common Mistakes
↑ Back to Contents- ❌ Treating like Y-tube (binary choice)
- ❌ Unequal airflow
- ❌ Not rotating odors
- ❌ Ignoring time-based data
- ❌ Overcrowding insects
📊 How Data is Recorded (Four-Way Specific)
↑ Back to Contents1. Key Concept
👉 Data = distribution, not decision
2. Example Data Table
| Insect | Arm A (sec) | Arm B (sec) | Arm C (sec) | Arm D (sec) | Entries A | B | C | D |
|---|---|---|---|---|---|---|---|---|
| 1 | 120 | 30 | 20 | 10 | 3 | 1 | 1 | 1 |
3. Interpretation
- Higher time = attraction
- More visits = exploratory interest
- No movement = discard
4. Important Rules
- ✔️ Entire observation counts
- ✔️ Return movements are INCLUDED
- ✔️ Multiple entries are meaningful
5. Duration Recording
- Continuous stopwatch OR
- Video tracking (preferred in literature)
6. Final Output
- % time per odor
- Mean ± SD
- Statistical significance
🔑 Quick Comparison (Y vs 4-Way)
↑ Back to Contents| Feature | Y-Tube | 4-Way |
|---|---|---|
| Output | Choice | Time distribution |
| Decision | One-time | Continuous |
| Stop rule | After choice | Full duration |
| Data type | Binary | Quantitative |
🔥 Practical Insight (Important)
↑ Back to ContentsMost beginners make this mistake:
👉 They try to force “choice” interpretation in 4-way system
This is wrong.
- ✔️ 4-way = behavioral intensity + preference gradient
- ✔️ Y-tube = decision test