🧪 Standard Operating Procedure (SOP)
Y-Tube Olfactometer Assay for Insect Behavior
1. Objective
↑ Back to ContentsTo evaluate insect behavioral response (attraction/repellency) to odor sources using a Y-tube olfactometer under controlled airflow conditions.
2. Apparatus & Materials
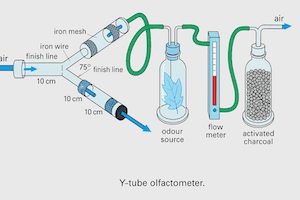
↑ Back to Contents- Y-tube olfactometer (glass/acrylic)
- एयर delivery system (pump + flow meters)
- Activated charcoal filters (for clean air)
- Humidifier / water wash bottles
- Odor source containers (glass chambers)
- Tubing (inert, e.g., PTFE/silicone)
- Insects (uniform age, sex if required)
- Stopwatch / timer
- Data recording sheet (your Excel sheet)
3. Pre-Experiment Setup
↑ Back to Contents3.1 Cleaning
- Wash Y-tube with neutral detergent → distilled water → ethanol
- Air dry completely
- Avoid residual odor contamination
3.2 Airflow Setup
- Maintain equal airflow in both arms
- Typical: 200–500 ml/min per arm
- Ensure:
- Smooth laminar flow
- No leakage
3.3 Odor Preparation
- Place:
- Test odor in one arm
- Control (clean air/solvent) in the other
- Randomize left/right placement
4. Experimental Conditions
↑ Back to Contents- Temperature: 25 ± 2°C
- Relative humidity: 60–80%
- Light: uniform, no directional bias
- Avoid external odors (perfume, chemicals)
5. Insect Preparation
↑ Back to Contents- Use healthy, active insects
- Standardize:
- Age
- Feeding status (e.g., starved 4–24 hrs depending on species)
- Acclimatize insects to lab conditions before testing
6. Procedure
↑ Back to ContentsStep 1: Start airflow
- Run clean air through system for 2–5 minutes before introducing insect
Step 2: Release insect
- Introduce a single insect at the base of the Y-tube
Step 3: Observation
- Allow insect to move freely
- Record behavior using defined criteria
7. Decision Criteria (VERY IMPORTANT)
↑ Back to Contents✔️ Valid Choice
- Insect crosses decision line (≈ 1/3–1/2 arm length)
- Remains oriented in that arm
❌ Not a Choice
- Insect stays at junction
- Moves slightly into arm and returns without crossing decision line
✔️ After crossing decision line
- Choice is final, even if insect comes back
8. Time Rules
↑ Back to Contents- Max observation time: 3–5 minutes per insect
- If no choice within time → record as:
- “No Choice” / Non-responder
Optional:
- Record time to first choice (seconds)
9. Replication
↑ Back to Contents- Minimum: 20–50 insects per treatment
- Perform:
- Multiple replicates
- On different days if possible
10. Bias Control
↑ Back to Contents- Switch odor arms after every 5–10 insects
- Rotate Y-tube (if possible)
- Run blank test (clean air vs clean air)
- Expect ~50:50 distribution
11. Data Recording
↑ Back to ContentsRecord for each insect:
- Choice (Left / Right / No Choice)
- Time to choice (optional)
- Notes (hesitation, unusual movement)
Use your prepared Excel sheet for:
- % choice
- Chi-square test
- Statistical validation
12. Data Analysis
↑ Back to Contents- Exclude or report non-responders separately
- Use:
- Chi-square test (Left vs Right)
- Significance:
- p < 0.05 → meaningful preference
13. Cleaning Between Runs
↑ Back to Contents- After 5–10 insects:
- Clean Y-tube OR
- At least flush with clean air
- Between treatments:
- Full cleaning required
14. Acceptance Criteria (How to know data is reliable)
↑ Back to Contents- ✔️ Control test ≈ 50:50 response
- ✔️ Consistent trend across replicates
- ✔️ Low % of non-responders (<30% ideal)
- ✔️ Stable airflow throughout experiment
15. Common Mistakes to Avoid
↑ Back to Contents- Unequal airflow ❌
- Recording choice at junction ❌
- Not switching odor sides ❌
- Using stressed or inactive insects ❌
- Contaminated glassware ❌
🔑 Quick Lab Summary
↑ Back to Contents- Decision = crossing marked line
- Time limit = 3–5 min
- Sample size = 20–50 insects
- Validate with control test + chi-square